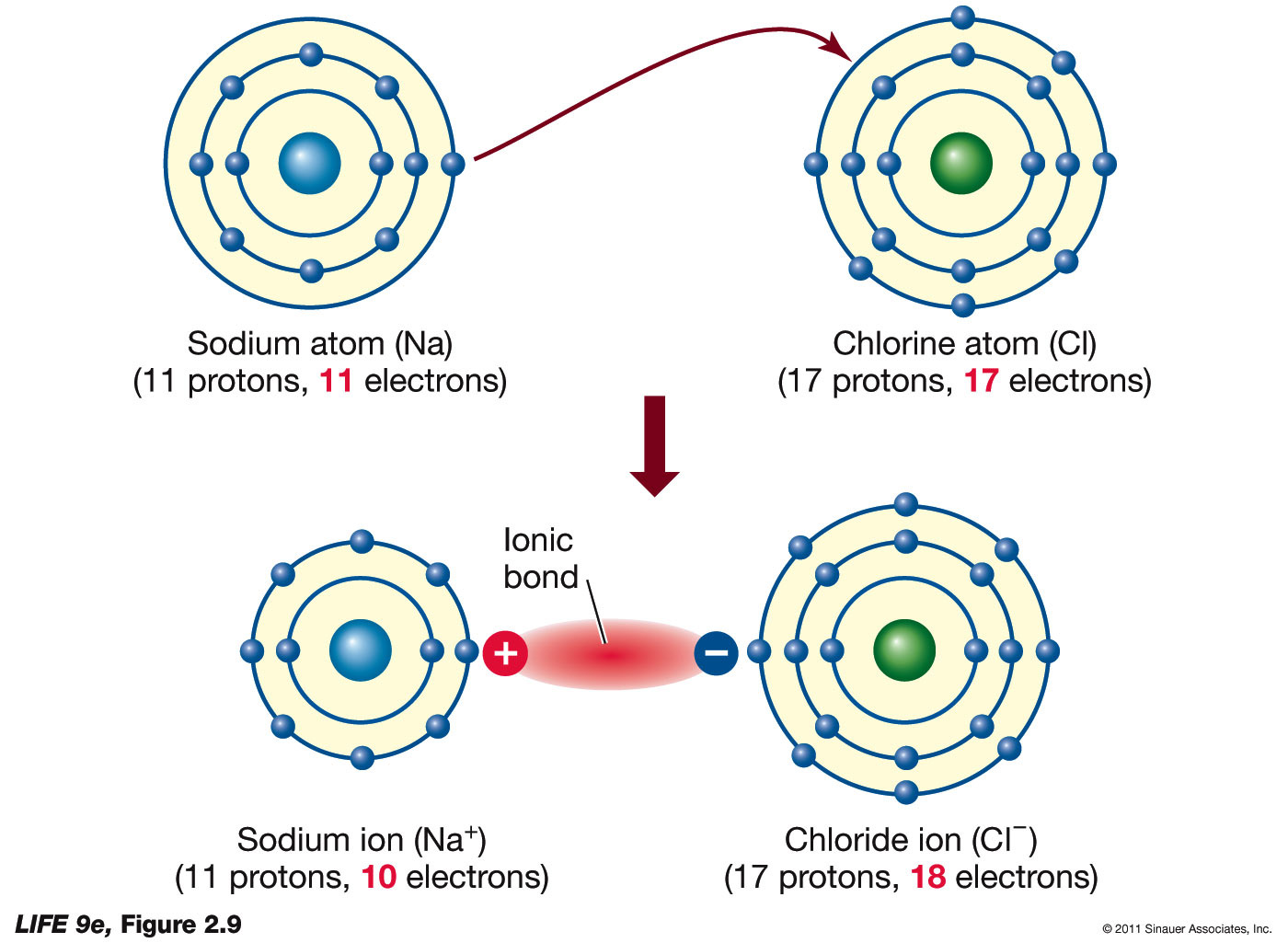
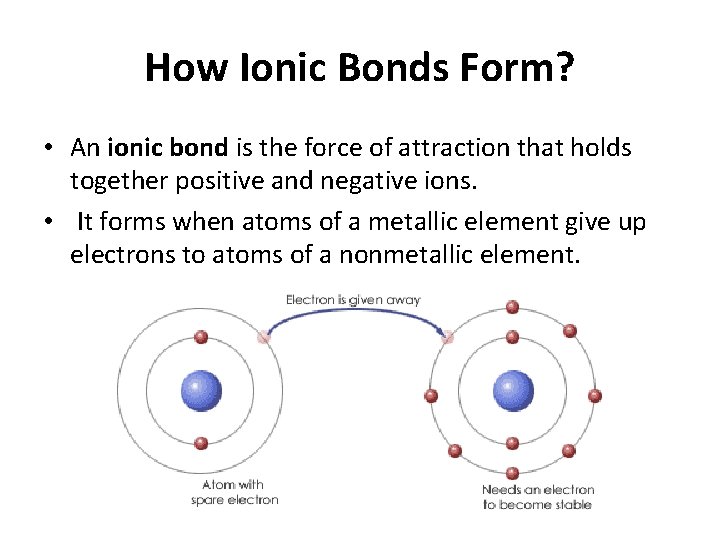
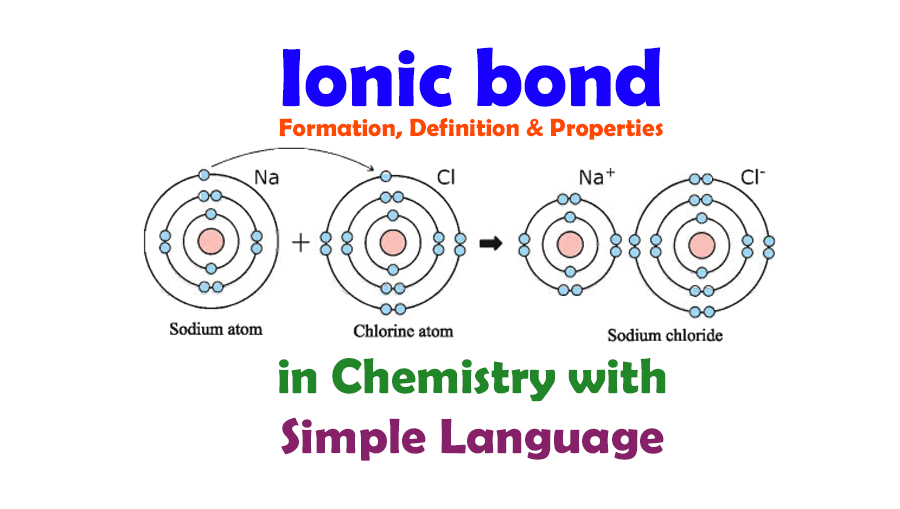
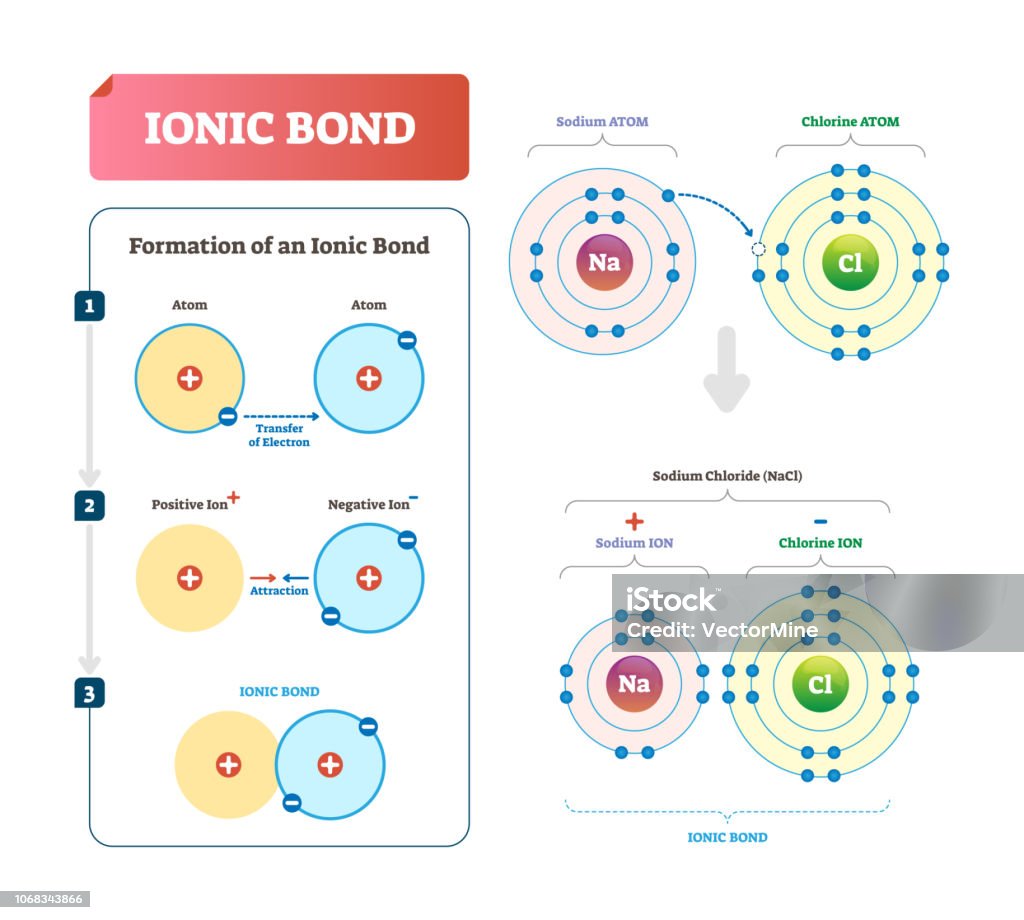
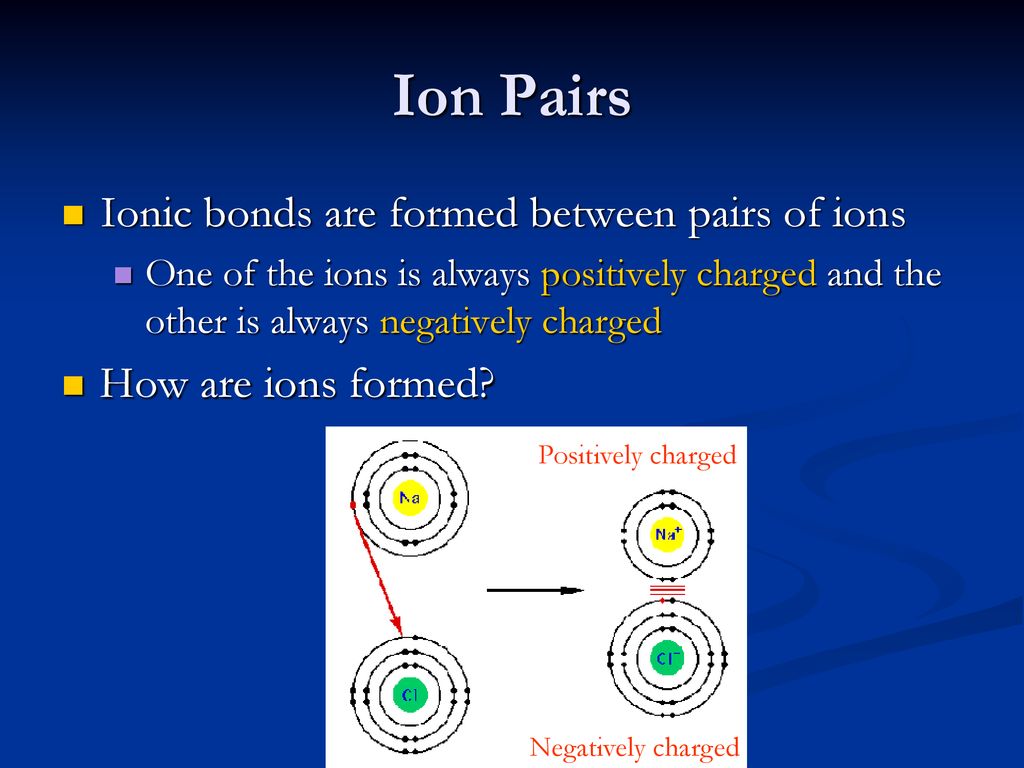
How Do Ions Form Ionic Bonds - Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Consider a single pair of ions,.
Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. Consider a single pair of ions,.
Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. Consider a single pair of ions,. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of. Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule.
Nomenclature & Chemical Bonding ppt download
Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. In this lesson, you'll discover.
Ionic Bonding Presentation Chemistry
The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. Consider a single pair of ions,. Ionic bonds are formed when positively and negatively.
ionic bond Definition, Properties, Examples, & Facts Britannica
Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Consider a single pair of ions,. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all.
Ionic Bond Formation Edexcel GCSE Chemistry Revision
The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and.
Diagram Of An Ion Ionic Compound Bond Examples Bonding Examp
Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of.
Ionic Bonds Essential Question How do ionic bonds
Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions..
Properties Of Ionic Compounds
Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule. Consider a single pair of ions,. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of. In this lesson, you'll discover how metals transfer.
Understanding Types of Chemical Bonds TEAS NurseHub
In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. Consider a single pair of ions,. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally.
Ion Definition
Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby.
Ionic Bond Vector Illustration Labeled Diagram With Formation
The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Ionic.
Consider A Single Pair Of Ions,.
Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. The attractive forces between ions are isotropic—the same in all directions—meaning that any particular ion is equally attracted to all of the nearby ions of. Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces.
.PNG)